AWS for Industries
AWS and Novartis: Re-inventing pharma manufacturing
Today, AWS announced a new strategic collaboration with Novartis that will transform Novartis’ core supply chain, manufacturing, and delivery operations through the use of AWS services. For several years, many AWS partners and life science customers have been using AWS services to modernize various aspects of manufacturing and supply chain. This strategic collaboration with Novartis is distinct by its broad strategic approach to transformation, and marks an acceleration of focused investment by AWS in this area. In this blog post, we will identify why manufacturing and supply chain transformations have become an urgent priority for pharmaceutical companies, and how AWS is uniquely positioned to assist customers along this journey. While the nature of the collaboration between Novartis and AWS spans supply chain, manufacturing, and distribution, the technical detail that follows will focus on the manufacturing portion. We will discuss how AWS is helping Novartis transform its manufacturing process by unifying access to all information and enabling Novartis to make quick and informed critical decisions.
Reinventing drug manufacturing and supply chains
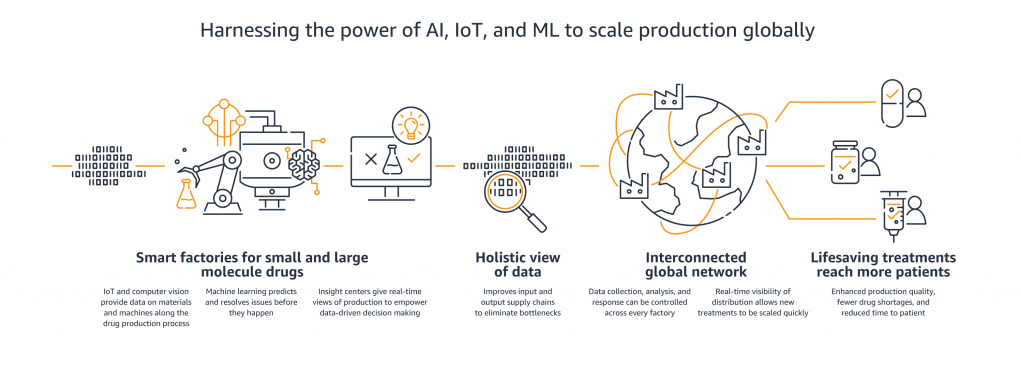
In recent years, targeted and more effective therapies and vaccines have been made possible by new biologic molecules. These therapies have had a positive impact on a broad range of diseases, including asthma, psoriasis, and various forms of cancer. These “large molecule” therapies can be more complex to manufacture, and can create new logistical challenges since they include living organisms. At the same time, pharmaceutical companies need to reduce the cost of manufacturing their “small molecule” therapies as those drugs lose patent protection, while needing to scale up production to address growing demand from developing markets, such as China. Moreover, the pharmaceutical manufacturing process has historically been focused on producing very large quantities of a particular therapy. Newer, genetically engineered therapies are designed for smaller cohorts, and may even be personalized down to an individual level. The combination of these challenges requires manufacturers to make their upstream supply chains, manufacturing processes, and downstream distribution more visible, predictable, efficient, and adaptable. For many of our customers the ultimate goal is an automated system that responds in real time to all relevant information. This is true not just at the factory level, but also at the enterprise level — managers want to see throughput data for all factories globally in order to ramp certain production lines up or down, thereby minimizing drug shortages and optimizing capacity.
Amazon has spent years developing extremely sophisticated supply chain and automation systems that enable millions of products to be delivered to hundreds of millions of individuals, within hours. These technologies and experiences uniquely position AWS to help companies reinvent their supply chain and production systems. For example, in March of this year AWS and Volkswagen announced a collaboration to transform automotive manufacturing. When Novartis inquired about a potential collaboration in manufacturing and supply chain, our AWS Life Science practice saw an opportunity to help a pharmaceutical industry leader positively impact the lives millions of patients.
Novartis is a large multinational medicines company, operating more than 60 manufacturing sites that produce therapeutics used by nearly 1 billion patients in 155 countries, annually. Like any modern manufacturer, site control systems provide operational metrics to site managers about the efficiency of individual machines, and facilitate day-to-day maintenance tasks. Shift handover is a mission critical point where information about operational performance is transferred from one crew to another. Engineers know production lines inside and out, but have lacked the operational metrics to support moving from anecdotal to data driven decision making. Across the Novartis Technical Operations (NTO) group, it has been difficult and costly to develop standard metrics for global site efficiency, see global operational performance in a “single pane of glass“, and build advanced machine learning models to predict site performance.
Prior to this collaboration, Novartis manufacturing site metrics had already been centralized into a traditional Hadoop-based big data platform, which has enabled the creation of extensive operational reporting. Data is periodically ingested from local historians into Hadoop Distributed File System (HDFS), where it is batch processed and prepared for reporting. While this has delivered value to the business, the operational reports are based upon fixed datasets which may be out of date in relation to operational decision-making. Due to the use of legacy third-party vendors, this batch mechanism has not scaled to meet the needs of the business, including: more users, additional metrics, advanced analytics and Machine Learning, and real-time data stream processing.
Through this collaboration, AWS and Novartis are jointly developing “Insight Centers” that provide real-time, interactive operational information to both site operators and corporate users around the globe. Insight Centers ingest site operational metrics through existing components such as local and global historians – platforms that capture industrial sensor data and add context and environmental meaning – as well as through new sensor integration. Existing historians will supply data from ‘brownfield’ sensors through nearly 20 SAP systems, while AWS IoT Greengrass edge devices will deliver richer data sources, such as images and video, and all data will be stored on Amazon S3. In addition to highly available and low cost object storage for this data, AWS IoT Core and IoT analytics provides data forwarding to an IoT optimized time series database where Novartis can perform real-time interpolation to facilitate predictive models.
Insight Centers will provide a cloud-native and ultra-scalable environment where existing big data processing technology can be run with minor changes due to compatibility with the most common frameworks and tools, but at lower cost, and enabling new uses cases that weren’t previously possible like computer vision powered line inspection. Daily production line & site operations will move to real-time data feeds, and add risk assessment models for proactive maintenance. Each site Insight Center will integrate with the others, providing a global view of the production capacity for each therapy. Finally, global Insight Center integration will deliver per product and process traceability, including manufacturing status, inventory levels, and cost.

As the volume, variety, and veracity of real-time data flow into the Insight Centers increases, the AWS AI/ML services will enable Novartis to create sophisticated ML models to advance its operational forecasting. The proposed technical architecture will create dynamic and flexible views of the Novartis global manufacturing processes — views that were previously un-achievable. Novartis will soon be able to fully leverage AWS’s machine learning and AI services to predict machine failure, develop digital twins, and build forecasting models of demand that will enable more efficient supply. These sophisticated use cases build upon the current initiatives already under way at Novartis. For example, Novartis is currently using Amazon SageMaker to build a computer vision-based model that will determine line clearance. Amazon SageMaker is a fully managed service that covers the entire machine learning workflow to label and prepare data, develop an algorithm, train the model, tune and optimize it for deployment, make predictions, and take action. In addition to Amazon SageMaker, AWS offers fully managed API-based AI services such as Amazon Textract and Amazon Comprehend. Novartis is also currently using these AI services to automate analysis of data extracted from printed manufacturing documents.
The Insight Centers will also be able to take advantage of AWS Identity & Access Management (IAM) to build a robust security posture and simplify entitlement management. IAM is a key co-traveler to data governance to ensure secure and appropriate sharing of manufacturing data among data scientists. Once approved, data scientists will have an open environment in which they can experiment with new supply chain and manufacturing optimization solutions that were previously constrained by legacy operating environment.
With these key features (and others to follow), Novartis Insight Centers will make quality manufacturing data available in real-time, and enable fast, informed decision-making. Improving the agility of the manufacturing process ultimately improves production quality while reducing cost, unnecessary inventory, and machine downtime. Transforming manufacturing will add value to Novartis operations, and will ultimately deliver the most important value to patients receiving Novartis therapies. This transformation roadmap will enable a future where small batch and even personalized treatments can be developed and manufactured faster, with higher quality, lower costs, and delivered on schedule.