AWS Big Data Blog
Improve clinical trial outcomes by using AWS technologies
We are living in a golden age of innovation, where personalized medicine is making it possible to cure diseases that we never thought curable. Digital medicine is helping people with diseases get healthier, and we are constantly discovering how to use the body’s immune system to target and eradicate cancer cells. According to a report published by ClinicalTrials.gov, the number of registered studies hit 293,000 in 2018, representing a 250x growth since 2000.
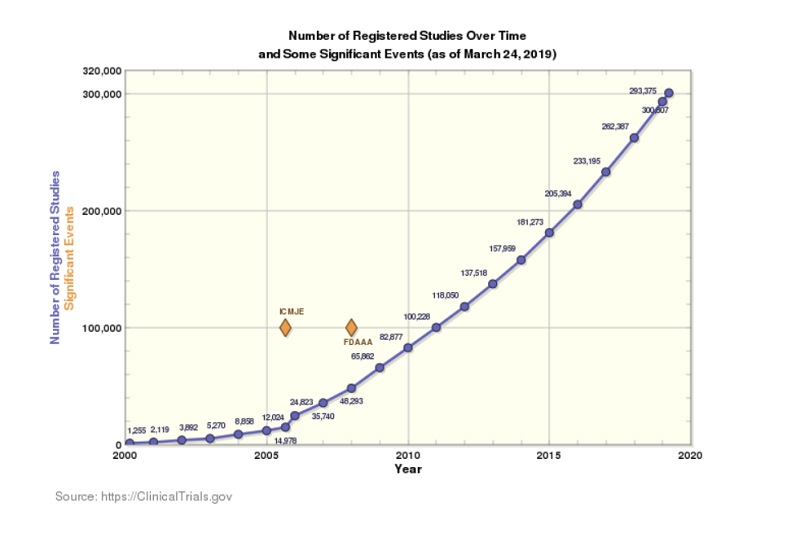
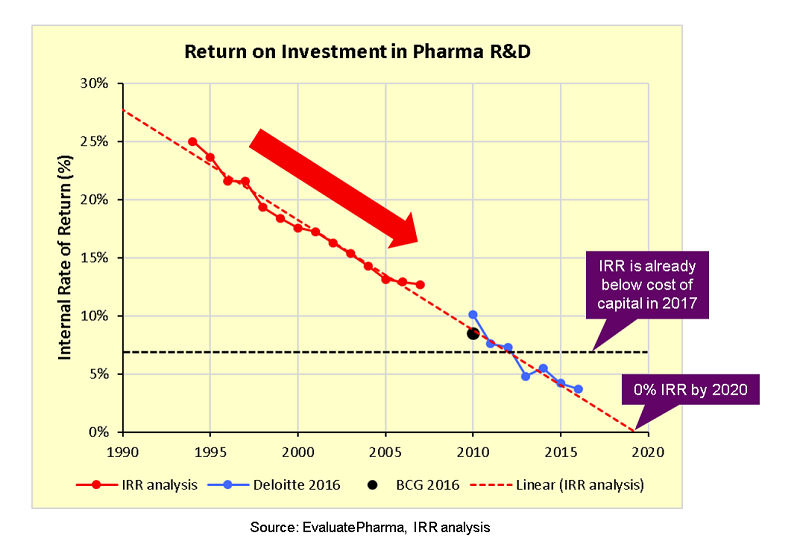
However, an internal rate of return (IRR) analysis by Endpoints News, using data from EvaluatePharma, highlights some interesting trends. A flourishing trend in pharma innovation is supported by strong growth in registered studies. However, the IRR shows a rapidly declining trend, from around 17 percent in 2000 to below the cost of capital in 2017 and projected to go to 0 percent by 2020.
This blog post is the first installment in a series that focuses on the end-to-end workflow of collecting, storing, processing, visualizing, and acting on clinical trials data in a compliant and secure manner. The series also discusses the application of artificial intelligence and machine learning technologies to the world of clinical trials. In this post, we highlight common architectural patterns that AWS customers use to modernize their clinical trials. These incorporate mobile technologies for better evidence generation, cost reduction, increasing quality, improving access, and making medicine more personalized for patients.
Improving the outcomes of clinical trials and reducing costs
Biotech and pharma organizations are feeling the pressure to use resources as efficiently as possible. This pressure forces them to search for any opportunity to streamline processes, get faster, and stay more secure, all while decreasing costs. More and more life sciences companies are targeting biologics, CAR-T, and precision medicine therapeutics, with focus shifting towards smaller, geographically distributed patient segments. This shift has resulted in an increasing mandate to capture data from previously unavailable, nontraditional sources. These sources include mobile devices, IoT devices, and in-home and clinical devices. Life sciences companies merge data from these sources with data from traditional clinical trials to build robust evidence around the safety and efficacy of a drug.
Early last year, the Clinical Trials Transformation Initiative (CTTI) provided recommendations about using mobile technologies for capturing holistic, high quality, attributable, real-world data from patients and for submission to the U.S. Food and Drug Administration (FDA). By using mobile technologies, life sciences companies can reduce barriers to trial participation and lower costs associated with conducting clinical trials. Global regulatory groups such as the FDA, Health Canada, and Medicines and Healthcare products Regulatory Agency (MHRA), among others, are also in favor of using mobile technologies. Mobile technologies can make patient recruitment more efficient, reach endpoints faster, and reduce the cost and time required to conduct clinical trials.
Improvised data ingestion using mobile technologies can speed up outcomes, reduce costs, and improve the accuracy of clinical trials. This is especially true when mobile data ingestion is supplemented with artificial intelligence and machine learning (AI/ML) technologies.
Together, they can usher in a new age of smart clinical trials.
At the same time, traditional clinical trial processes and technology designed for mass-marketed blockbuster drugs can’t effectively meet emerging industry needs. This leaves life sciences and pharmaceutical companies in need of assistance for evolving their clinical trial operations. These circumstances result in making clinical trials one of the largest areas of investment for bringing a new drug to market.
Using mobile technologies with traditional technologies in clinical trials can improve the outcomes of the trials and simultaneously reduce costs. Some of the use cases that the integration of various technologies enables include these:
- Identifying and tracking participants in clinical trials
- Identifying participants for clinical trials recruitment
- Educating and informing patients participating in clinical trials
- Implementing standardized protocols and sharing associated information to trial participants
- Tracking adverse events and safety profiles
- Integrating genomic and phenotypic data for identifying novel biomarkers
- Integrating mobile data into clinical trials for better clinical trial management
- Creation of a patient-control arm based on historical data
- Stratifying cohorts based on treatment, claims, and registry datasets
- Building a collaborative, interoperable network for data sharing and knowledge creation
- Building compliance-ready infrastructure for clinical trial management
The AWS Cloud provides HIPAA eligible services and solutions. As an AWS customer, you can use these to build solutions for global implementation of mobile devices and sensors in trials, secure capture of streaming Internet of Things (IoT) data, and advanced analytics through visualization tools or AI/ML capabilities. Some of the use cases these services and solutions enable are finding and recruiting patients using smart analytics, facilitating global data management, and remote or in-patient site monitoring. Others include predicting lack of adherence, detecting adverse events, and accelerating trial outcomes along with optimizing trial costs.
Clinical Trials 2.0 (CT2.0) at AWS is geared toward facilitating wider adoption of cloud-native services to enable data ingestion from disparate sources, cost-optimized and reliable storage, and holistic analytics. At the same time, CT2.0 provides the granular access control, end-to-end security, and global scalability needed to conduct clinical trials more efficiently.
Reference architecture
One of the typical architectures for managing a clinical trial using mobile technologies is shown following. This architecture focuses on capturing real-time data from mobile sources and providing a way to process it.
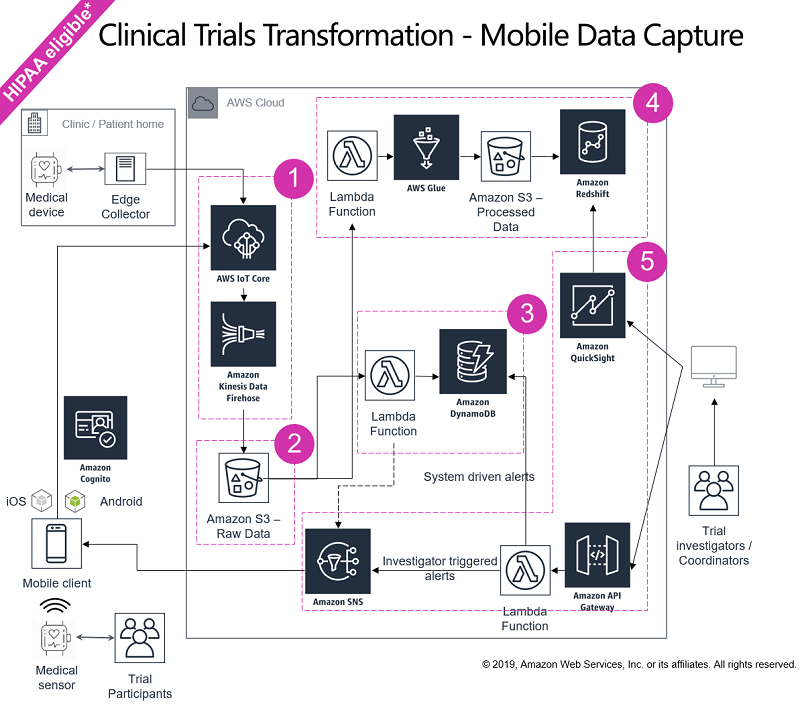
* – Additional considerations such as data security, access control, and compliance need to be incorporated into the architecture and are discussed in the remainder of this post.
Managing a trial by using this architecture consists of the following five major steps.
Step 1: Collect data
Mobile devices, personal wearables, instruments, and smart-devices are extensively being used (or being considered) by global pharmaceutical companies in patient care and clinical trials to provide data for activity tracking, vital signs monitoring, and so on, in real-time. Devices like infusion pumps, personal use dialysis machines, and so on require tracking and alerting of device consumables and calibration status. Remote settings management is also a major use case for these kinds of devices. The end-user mobile devices used in the clinical trial emit a lot of telemetry data that requires real-time data capture, data cleansing, transformation, and analysis.
Typically, these devices are connected to an edge node or a smart phone. Such a connection provides sufficient computing resources to stream data to AWS IoT Core. AWS IoT Core can then be configured to write data to Amazon Kinesis Data Firehose in near real time. Kinesis Data Firehose is a fully managed service for delivering real-time streaming data to destinations such as Amazon S3. S3 provides online, flexible, cost efficient, pay-as-you-go storage that can replicate your data on three Availability Zones within an AWS Region. The edge node or smart phone can use the AWS IoT SDKs to publish and subscribe device data to AWS IoT Core with MQTT. This process uses the AWS method of authentication (called ‘SigV4’), X.509 certificate–based authentication, and customer-created token-based authentication (through custom authorizers). This authenticated approach enables you to map your choice of policies to each certificate and remotely control device or application access. You can also use the Kinesis Data Firehose encryption feature to enable server-side data encryption.
You can also capture additional data such as Case Report Forms (CRF), Electronic Medical Records (EMR), and medical images using Picture Archiving and Communication Systems (PACS). In addition, you can capture laboratory data (Labs) and other Patient Reported Outcomes data (ePRO). AWS provides multiple tools and services to effectively and securely connect to these data sources, enabling you to ingest data in various volumes, variety, and velocities. For more information about creating a HealthCare Data Hub and ingesting Digital Imaging and Communications in Medicine (DICOM) data, see the AWS Big Data Blog post Create a Healthcare Data Hub with AWS and Mirth Connect.
Step 2: Store data
After data is ingested from the devices and wearables used in the clinical trial, Kinesis Data Firehose is used to store the data on Amazon S3. This stored data serves as a raw copy and can later be used for historical analysis and pattern prediction. Using Amazon S3’s lifecycle policies, you can periodically move your data to reduced cost storage such as Amazon S3 Glacier for further optimizing their storage costs. Using Amazon S3 Intelligent Tiering can automatically optimize costs when data access patterns change, without performance impact or operational overhead by moving data between two access tiers—frequent access and infrequent access. You can also choose to encrypt data at rest and in motion using various encryption options available on S3.
Amazon S3 offers an extremely durable, highly available, and infinitely scalable data storage infrastructure, simplifying most data processing, backup, and replication tasks.
Step 3: Data processing—fast lane
After collecting and storing a raw copy of the data, Amazon S3 is configured to publish events to AWS Lambda and invoke a Lambda function by passing the event data as a parameter. The Lambda function is used to extract the key performance indicators (KPIs) such as adverse event notifications, medication adherence, and treatment schedule management from the incoming data. You can use Lambda to process these KPIs and store them in Amazon DynamoDB, along with encryption at rest, which powers a near-real-time clinical trial status dashboard. This alerts clinical trial coordinators in real time so that appropriate interventions can take place.
In addition to this, using a data warehouse full of medical records, you can train and implement a machine learning model. This model can predict which patients are about to switch medications or might exhibit adherence challenges in the future. Such prediction can enable clinical trial coordinators to narrow in on those patients with mitigation strategies.
Step 4: Data processing—batch
For historical analysis and pattern prediction, the staged data (stored in S3) is processed in batches. A Lambda function is used to trigger the extract, transform, and load (ETL) process every time new data is added to the raw data S3 bucket. This Lambda function triggers an ETL process using AWS Glue, a fully managed ETL service that makes it easy for you to prepare and load your data for analytics. This approach helps in mining current and historical data to derive actionable insights, which is stored on Amazon S3.
From there, data is loaded on to Amazon Redshift, a cost-effective, petabyte-scale data warehouse offering from AWS. You can also use Amazon Redshift Spectrum to extend data warehousing out to exabytes without loading any data to Amazon Redshift, as detailed in the Big Data blog post Amazon Redshift Spectrum Extends Data Warehousing Out to Exabytes—No Loading Required. This enables you to provide an all-encompassing picture of the entire clinical trial to your clinical trial coordinators, enabling you to react and respond faster.
In addition to this, you can train and implement a machine learning model to identify patients who might be at risk for adherence challenges. This enables clinical trial coordinators to reinforce patient education and support.
Step 5: Visualize and act on data
After the data is processed and ready to be consumed, you can use Amazon QuickSight, a cloud-native business intelligence service from AWS that offers native Amazon Redshift connectivity. Amazon QuickSight is serverless and so can be rolled out to your audiences in hours. You can also use a host of third-party reporting tools, which can use AWS-supplied JDBC or ODBC drivers or open-source PostgreSQL drivers to connect with Amazon Redshift. These tools include TIBCO Spotfire Analytics, Tableau Server, Qlik Sense Enterprise, Looker, and others. Real-time data processing (step 3 preceding) combines with historical-view batch processing (step 4). Together, they empower contract research organizations (CROs), study managers, trial coordinators, and other entities involved in the clinical trial journey to make effective and informed decisions at a speed and frequency that was previously unavailable. Using Amazon QuickSight’s unique Pay-per-Session pricing model, you can optimize costs for your bursty usage models by paying only when users access the dashboards.
Using Amazon Simple Notification Service (Amazon SNS), real-time feedback based on incoming data and telemetry is sent to patients by using text messages, mobile push, and emails. In addition, study managers and coordinators can send Amazon SNS notifications to patients. Amazon SNS provides a fully managed pub/sub messaging for micro services, distributed systems, and serverless applications. It’s designed for high-throughput, push-based, many-to-many messaging. Alerts and notifications can be based on current data or a combination of current and historical data.
To encrypt messages published to Amazon SNS, you can follow the steps listed in the post Encrypting messages published to Amazon SNS with AWS KMS, on the AWS Compute Blog.
Data security, data privacy, data integrity, and compliance considerations
At AWS, customer trust is our top priority. We deliver services to millions of active customers, including enterprises, educational institutions, and government agencies in over 190 countries. Our customers include financial services providers, healthcare providers, and governmental agencies, who trust us with some of their most sensitive information.
To facilitate this, along with the services mentioned earlier, you should also use AWS Identity and Access Management (IAM) service. IAM enables you to maintain segregation of access, fine-grained access control, and securing end user mobile and web applications. You can also use AWS Security Token Service (AWS STS) to provide secure, self-expiring, time-boxed, temporary security credentials to third-party administrators and service providers, greatly strengthening your security posture. You can use AWS CloudTrail to log IAM and STS API calls. Additionally, AWS IoT Device Management makes it easy to securely onboard, organize, monitor, and remotely manage IoT devices at scale.
With AWS, you can add an additional layer of security to your data at rest in the cloud. AWS provides scalable and efficient encryption features for services like Amazon EBS, Amazon S3, Amazon Redshift, Amazon SNS, AWS Glue, and many more. Flexible key management options, including AWS Key Management Service, enable you to choose whether to have AWS manage the encryption keys or to keep complete control over their keys. In addition, AWS provides APIs for you to integrate encryption and data protection with any of the services that you develop or deploy in an AWS environment.
As a customer, you maintain ownership of your data, and select which AWS services can process, store, and host the content. Generally speaking, AWS doesn’t access or use customers’ content for any purpose without their consent. AWS never uses customer data to derive information for marketing or advertising.
When evaluating the security of a cloud solution, it’s important that you understand and distinguish between the security of the cloud and security in the cloud. The AWS Shared Responsibility Model details this relationship.
To assist you with your compliance efforts, AWS continues to add more services to the various compliance regulations, attestations, certifications, and programs across the world. To decide which services are suitable for you, see the services in scope page.
You can also use various services like, but not limited to, AWS CloudTrail, AWS Config, Amazon GuardDuty, and AWS Key Management Service (AWS KMS) to enhance your compliance and auditing efforts. Find more details in the AWS Compliance Solutions Guide.
Final thoughts
With the ever-growing interconnectivity and technological advances in the field of medical devices, mobile devices and sensors can improve numerous aspects of clinical trials. They can help in recruitment, informed consent, patient counseling, and patient communication management. They can also improve protocol and medication adherence, clinical endpoints measurement, and the process of alerting participants on adverse events. Smart sensors, smart mobile devices, and robust interconnecting systems can be central in conducting clinical trials.
Every biopharma organization conducting or sponsoring a clinical trial activity faces the conundrum of advancing their approach to trials while maintaining overall trial performance and data consistency. The AWS Cloud enables a new dimension for how data is collected, stored, and used for clinical trials. It thus addresses that conundrum as we march towards a new reality of how drugs are brought to market. The AWS Cloud abstracts away technical challenges such as scaling, security, and establishing a cost-efficient IT infrastructure. In doing so, it allows biopharma organizations to focus on their core mission of improving patent lives through the development of effective, groundbreaking treatments.
About the Author
 Mayank Thakkar – Global Solutions Architect, AWS HealthCare and Life Sciences
Mayank Thakkar – Global Solutions Architect, AWS HealthCare and Life Sciences
 Deven Atnoor, Ph.D. – Industry Specialist, AWS HealthCare and Life Sciences
Deven Atnoor, Ph.D. – Industry Specialist, AWS HealthCare and Life Sciences