Life Sciences Resources
Events
Learn more about upcoming Life Sciences events and connect with others in your industry.
Explore Life Sciences events
Videos & Webinars



Whitepapers & Briefs
Reference Architectures
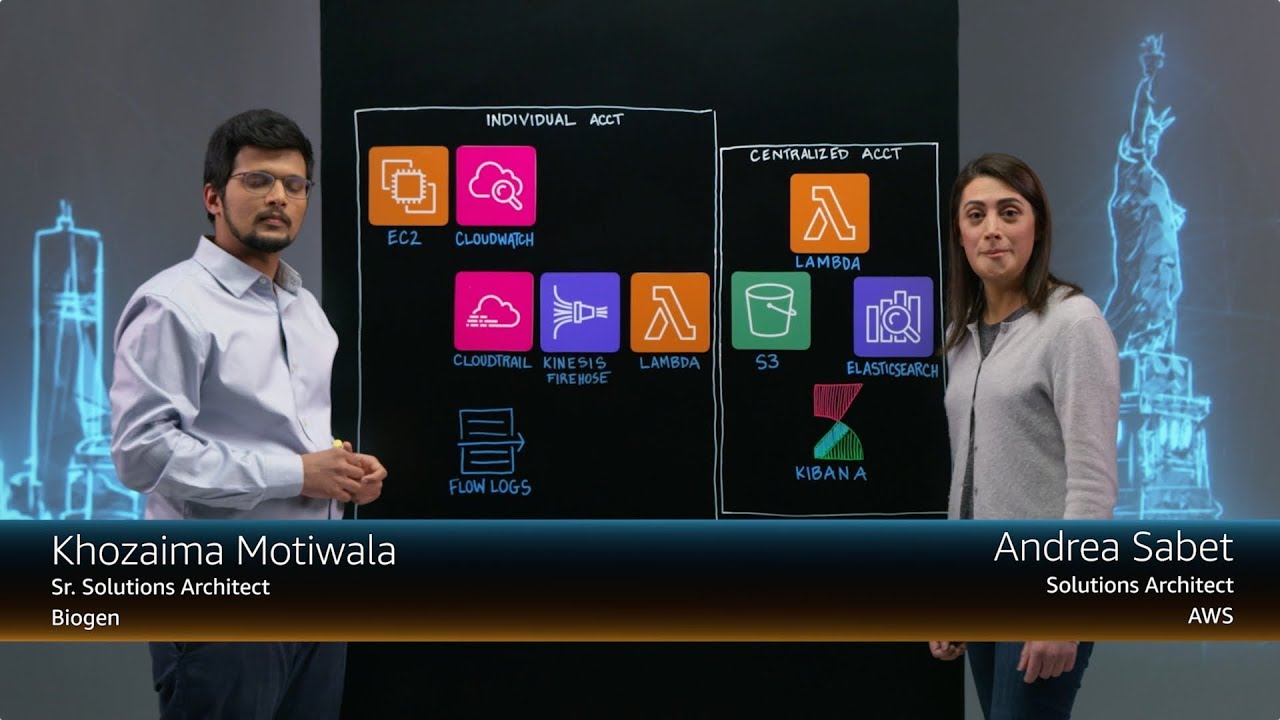
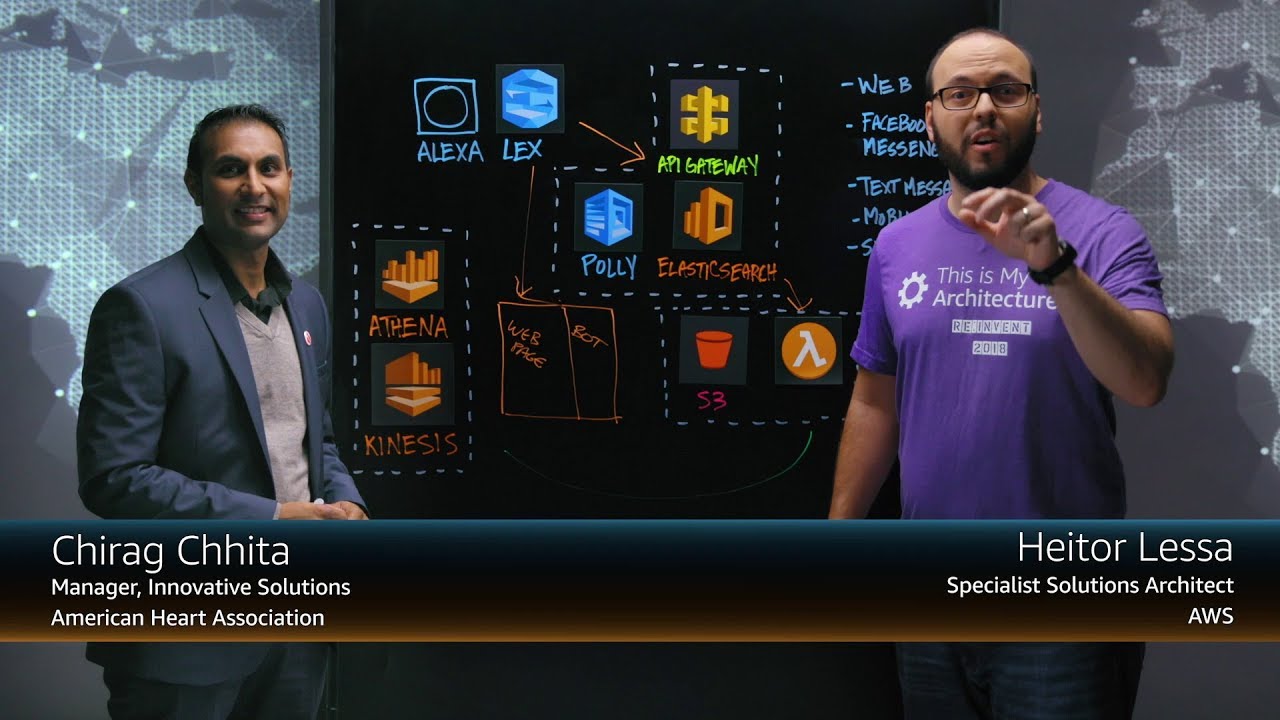
Case Studies
Merck used AWS to build the Change Assessment Knowledge Engine (CAKE). CAKE is powered by an Amazon Neptune graph database containing information about Merck’s supply chain and regulatory operating environment. CAKE reduced change assessment duration by up to 90 percent and total manual effort per assessment by 30–70 percent.
 Learn more »
Learn more »
Using AWS, biopharmaceutical company AstraZeneca built a cloud-based, efficient, scalable solution that processes genomics sequencing data quickly.
 Learn more
Learn more
Biotech company Moderna Therapeutics, along with its partners, initiated clinical trials for seven drugs—with nine more in the pipeline—by taking advantage of AWS computing power, integration, and scalability. Moderna operates differently from both traditional pharmaceutical companies and biotech drug companies by delivering vaccines and therapies via messenger RNA.
 Learn more »
Learn more »
Online health community Inspire used Amazon SageMaker to connect patients with relevant content on its platform of 1.5 billion words written by Inspire members representing 3,600+ medical conditions.
 Learn more »
Learn more »
Scientists and lab managers are challenged to accelerate their productivity, optimize processes, and prevent errors while performing wet lab research. Thermo Fisher Scientific (Thermo Fisher) is using Amazon Web Services (AWS) tools to equip labs to aid scientists and lab managers in this work.
 Learn more
Learn more
AWS products allow life sciences startups to compete with Big Pharma by using cloud services. Idorsia is a startup pharmaceutical company that discovers and develops innovative medicines. Idorsia runs 90 percent of its computing needs using AWS products such as Amazon WorkSpaces and AWS Lambda.
 Learn more »
Learn more »
Get started
Leading life sciences companies are already using AWS. Contact our experts and start your own AWS Cloud journey today.
Contact Sales
Did you find what you were looking for today?
Let us know so we can improve the quality of the content on our pages
