AWS Public Sector Blog
How Servizo Galego de Saúde accelerates clinical trials with AWS
Spain leads Europe in clinical trials, with over 930 authorized studies in 2024 and recruitment rates up to 40% faster than the European average in oncology. However, the European healthcare sector faces critical challenges. Data fragmentation, patient recruitment bottlenecks, and regulatory complexity threaten global competitiveness. Between 2018 and 2023, approximately 60,000 fewer European patients gained access to experimental treatments because of these inefficiencies.
The Servizo Galego de Saúde (SERGAS) has taken a decisive step to address these challenges with Innovatrial, a next-generation digital offering built on Research and Engineering Studio (RES) on Amazon Web Services (AWS) by NTT DATA in collaboration with AWS. This solution radically transforms how clinical trials are managed, connecting data, people, and processes in a unified environment that accelerates scientific evidence generation.
Fragmentation in a complex environment
Traditional clinical trials operate in silos. Patient data is scattered across incompatible hospital systems, researchers lack methods to quickly identify eligible candidates, and ethics committees rely on manual review processes that delay approvals. This fragmentation increases operational costs and undermines efficiency across Europe’s clinical research environment.
The European Medicines Agency‘s vision for Clinical Evidence 2030 demands a fundamental shift: patient-centered trials, use of real-world data (RWD), and greater interoperability. To meet this agenda, healthcare institutions need digital infrastructure that deftly navigates technical and regulatory barriers.
Innovatrial running on RES on AWS
Innovatrial is a trusted research environment (TRE) designed specifically for biomedical research. It’s been implemented on RES on AWS by NTT DATA. This solution provides a more secure, scalable, collaborative environment where researchers, sponsors, contract research organizations (CROs), and regulators can work with sensitive data without compromising patient privacy.
The solution, developed and validated under a pre-commercial procurement contract in collaboration with the Centre for the Development of Industrial Technology (CDTI) – a public entity under the Ministry of Science, Innovation and Universities – SERGAS, and the associated research foundations, represents an innovative achievement that is ready for the upcoming deployment and implementation phase.
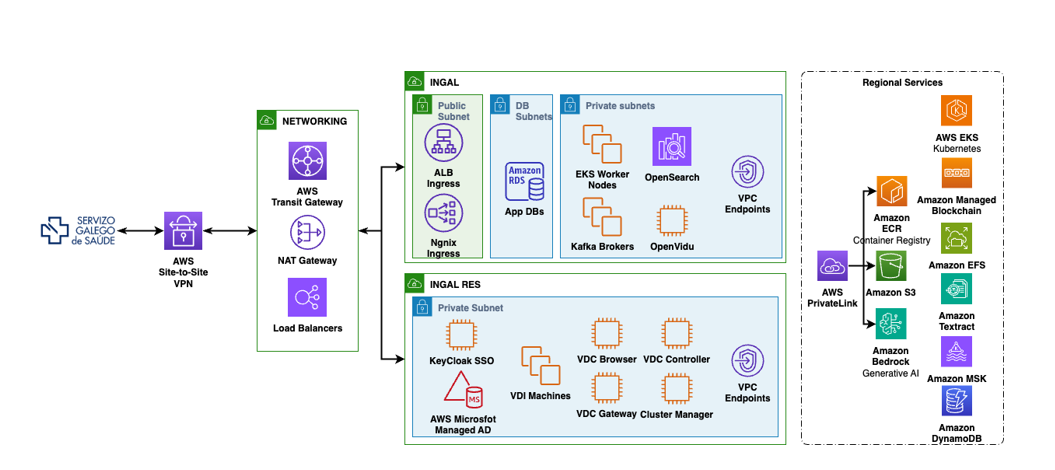
Architecture built on RES on AWS
Innovatrial’s architecture uses multiple AWS services to create a robust environment.
As foundation, InnovaTrial uses Amazon Simple Storage Service (Amazon S3) as a centralized data lake, storing both structured and unstructured data including medical images, genomic data, and clinical records. AWS Glue catalogs and transforms this data, while Amazon Athena enables direct SQL queries without requiring data movement between systems, creating a seamless data infrastructure for clinical research operations.
The platform implements global standards for interoperability, including HL7/FHIR for clinical data exchange, Clinical Data Interchange Standards Consortium (CDISC) for regulatory submissions, DICOM for real-world data integration, and IHE profiles for cross-institutional communication within medicine and biology. This adherence to standards ensures data flows frictionlessly between hospitals, research systems, and regulatory authorities—facilitating collaboration across the entire clinical trials ecosystem.
InnovaTrial uses Amazon SageMaker as the infrastructure to run NTT DATA’s custom machine learning models. These customer-developed models support patient cohort identification workflows using natural language processing. Researchers can formulate inclusion/exclusion criteria in natural language, and the system translates these queries into searchable OMOP databases, generating candidate cohorts in minutes instead of weeks. This AI-driven approach dramatically accelerates patient recruitment timelines.
Security and regulatory compliance are core pillars of the platform. AWS Identity and Access Management (IAM) implements role-based access controls so that each user accesses only necessary information. Amazon CloudWatch and AWS CloudTrail provide immutable audit logs, complying with GDPR, ICH-GCP, FDA CFR Title 21 Part 11, and EMEA guidelines. Data anonymization and pseudonymization follow the European Cybersecurity Act recommendations, protecting patient confidentiality while preserving scientific utility.
The platform provides isolated research environments on AWS where researchers can run analyses in secure, virtual workspaces. Amazon Elastic Compute Cloud (Amazon EC2) and AWS Batch provide the computational infrastructure for intensive workloads, supporting customer applications ranging from genomic analysis tools to trial simulation software. Additionally, Amazon Managed Blockchain facilitates trial data immutability, creating a permanent and transparent record that enhances regulatory and scientific trust throughout the research lifecycle.
The following diagram illustrates a multi-layered approach focused on security and environment isolation:
Accelerating clinical trial operations and research workflows
Innovatrial generates tangible benefits for stakeholders across the clinical research environment. For investigators, automated patient identification reduces recruitment time by weeks. Real-time alerts notify investigators when new patients meet eligibility criteria, keeping recruitment dynamic throughout the study life cycle. NTT’s Deep learning models learn from historical patterns to recommend cohorts, minimizing selection bias.
Real-time data monitoring means sponsors and CROs don’t need to rely on periodic manual reports. Dashboards powered by Amazon Quick Sight provide instant visibility into study progress, facilitating proactive protocol adjustments and resource allocation. Verified data collaboration accelerates evidence-based decision-making.
For ethics committees, centralization of study documents and data in a more secure location can drastically reduce review times. Automated workflows track regulatory milestones and documentation in real time, minimizing manual oversight.
With Innovatrial’s flexible architecture, decentralized and hybrid trial designs can expand access to research. Patients can participate from their homes through remote monitoring and applications integrated with healthcare services, lowering logistical barriers. Personalized communication and control over their data fosters patient trust and encourages long-term engagement.
Findable, Accessible, Interoperable, and Reusable (FAIR) principles maximize data value
Innovatrial operationalizes FAIR principles to maximize the scientific value of research data:
- Findable – Standardized metadata tagging and persistent identifiers make datasets traceable across studies
- Accessible – Access control layers mean authorized users can more securely access relevant information
- Interoperable – Adherence to global standards enables cross-study comparisons and integration with health data networks such as the Data Analysis and Real World Interrogation Network (DARWIN EU)
- Reusable – Curation and documentation protocols mean data remains valuable for secondary analyses and future research
A centralized terminology server manages internationally recognized clinical vocabularies such as Systematized Nomenclature of Medicine – Clinical Terms (SNOMED CT) and International Classification of Diseases, Tenth Revision (ICD-10), facilitating semantic consistency and reducing risk of misinterpretation.
Sustainability and scalability
Innovatrial’s digital-first approach contributes to financial and environmental sustainability. Automation of administrative tasks reduces operational costs and minimizes paper use, aligning with eco-friendly practices in the healthcare sector. AWS serverless architecture and managed services obviate the need to maintain physical infrastructure so SERGAS can focus on science rather than IT management.
The elastic scalability of AWS means Innovatrial can grow from local pilot studies to multinational collaborative networks without redesigning the architecture. As new hospitals and research centers join the service, computational and storage resources automatically adjust, maintaining performance and security.
Key trends for Innovatrial
- Decentralized and hybrid trials to expand access and inclusivity
- Advanced therapies and precision medicine requiring adaptive designs and integrated Real World Data (RWD)
- Rare disease research with smaller cohorts and federated analytics
- Primary care research networks that bring studies closer to communities
- Greater transparency through ready for the Clinical Trials Information System (CTIS), standards-based, machine-readable evidence
Looking to the future
Innovatrial positions SERGAS and Spain at the forefront of digital transformation in clinical research. As trials evolve toward advanced therapies, smaller populations, and distributed models, the solution provides a future-proof digital foundation for generating high-quality evidence efficiently and responsibly.
Organizations adopting this approach can shorten time-to-insight, reduce development risks, and deliver innovative therapies to patients faster. In an environment where European competitiveness in clinical research is at stake, Innovatrial demonstrates how cloud technology can turn systemic challenges into strategic advantages.
The collaboration between SERGAS, AWS, and NTT DATA exemplifies the power of public sector innovation. When healthcare institutions combine clinical expertise with advanced technological infrastructure, the result is faster, more transparent, more patient-centered science. Innovatrial is not only a solution, it’s a catalyst for the future of evidence-based medicine.
Additional resources
RES on AWS is an open source solution that research organizations can use to create and manage more secure, collaborative research environments in the cloud. It provides isolated virtual workspaces, granular access controls, and high-performance computing offerings. With RES on AWS, researchers can work with sensitive data while meeting strict security and regulatory compliance requirements.