AWS for Industries
Setting the “Industry 4.0” foundations for life sciences manufacturing
Introduction
This blog summarizes challenges in life sciences manufacturing along with examples of Amazon Web Services (AWS) customers using cloud technology to improve their operations. We explore reference architectures illustrating how AWS services can be used to establish the foundation for “Industry 4.0.” Finally, we describe an AWS assessment engagement customers can use to understand their digital manufacturing maturity, and we highlight a blog post on GxP allow lists, which are critical for bringing GxP workloads common in life sciences manufacturing environments to the cloud.
Assessing the potential and the challenges of life sciences manufacturing
The biopharma industry is in the beginning stages of its fourth revolution, commonly referred to across multiple industries as “Industry 4.0.”
To compound the need for change, current circumstances have introduced unforeseen challenges to life sciences companies’ ability to ensure supply. These challenges have affected upstream supply, worker safety, and resulted in unforeseen spikes in product demand.
Potential weaknesses have created acute pain, such as the overwhelming industry reliance on India for generics and on China for the supply of Active Pharmaceutical Ingredients (APIs).
With the US market specifically relying on China to supply 90% of its APIs, a large swath of global life sciences customers including Pfizer, Johnson and Johnson, and Novartis—and more specialist organizations like Vertex and Jazz Pharma—have been impacted. These impacts range from calcium supplements to HIV drugs and cancer therapies.
Simultaneously, demands for manufacturing agility and performance are increasing with the proliferation of precision medicines driven by scientific innovations in cellular and gene therapy. These new products are transforming manufacturing and supply chain models from the push-based models of the blockbuster era.
In light of these pressures and opportunities, forward-thinking companies are using technology, data analytics, and AWS to modernize their manufacturing operations.
For example, Moderna, a pioneer for mRNA therapeutics, has chosen AWS to power its highly automated production facility and supply chain.
Along with the digitization of supply chains, the industry is reaching an inflection point in how biopharma manufacturing modernizes production.
Novartis chose a strategic collaboration with AWS to accelerate its digital transformation journey in manufacturing and supply chain operations and management, setting the foundation to deliver enterprise-wide value by leveraging via data and analytics.
AWS has established itself as the backbone of these initiatives based on its experience powering Amazon’s industry-leading supply chain, a culture of innovation, and the use of microservices. Now, biopharma customers can establish a solid foundation for digital plant maturity and shift the industry towards becoming predictive and adaptive.
To streamline and accelerate the modernization of biopharmaceutical manufacturing, this blog outlines a prescriptive approach including a reference architecture with sequenced steps, tangible use cases that deliver measurable value, and ways to get started with AWS.
Foundations for digital plant maturity
The following reference architecture details the use of example AWS services through various phases. The reference architecture describes specific phases for the following:
- Establish connectivity
- Establish a cloud data lake
- Implement predictive analytics
- Implement use case: Alerting and real-time monitoring
Establish connectivity
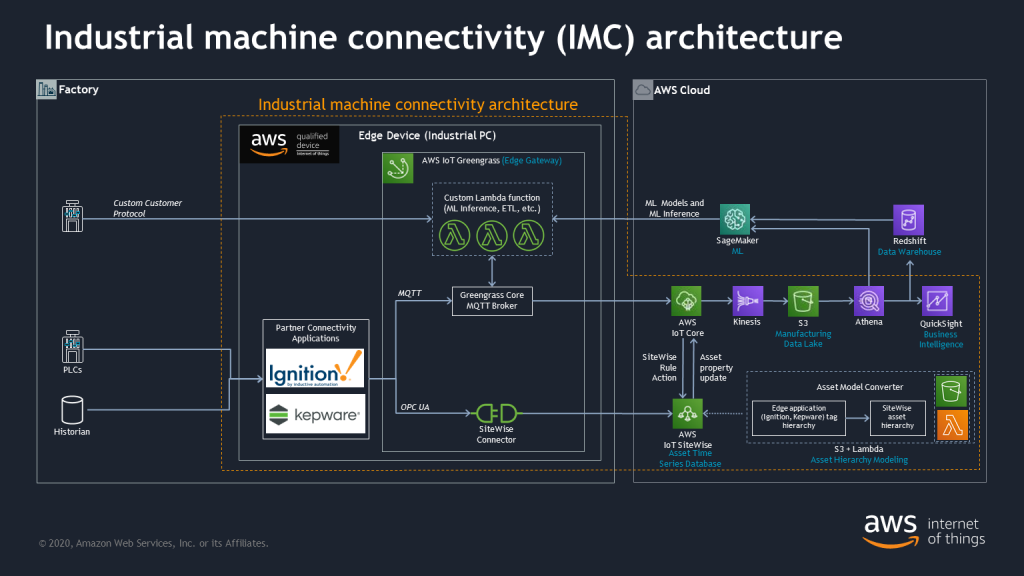
The first part of this reference architecture involves ingesting data from the shop floor.
Ingest: Ingest ensures that system data inter-operability is standardized such that data can flow freely and uniformly upstream and be readily available to individuals on the shop floor. The primary technical outcome employs a loosely coupled microservices architecture, which supports low-level sensors or specific instrument unit operations, standard transport protocols, and can consume multi-format/multi type data payloads. Ingest also includes the automation and digitization of paper-based workflows. All data flows are brokered by a common edge gateway mechanism.
To accelerate this first-mile step, AWS recently launched the industrial machine connectivity (IMC) kit. The IMC kit is an industry-agnostic solution designed to enable customers and partners to get data from their assets into the AWS Cloud in a simple, structured process so they can rapidly realize the business value that is derived from that data. Customers can convert existing asset hierarchy definitions (such as factory, lines, machines, tags, etc.) in partner edge applications like Inductive Automation’s Ignition Server or PTC’s KEPServerEX to the equivalent asset hierarchy within AWS IoT SiteWise.
Establishing a cloud data lake
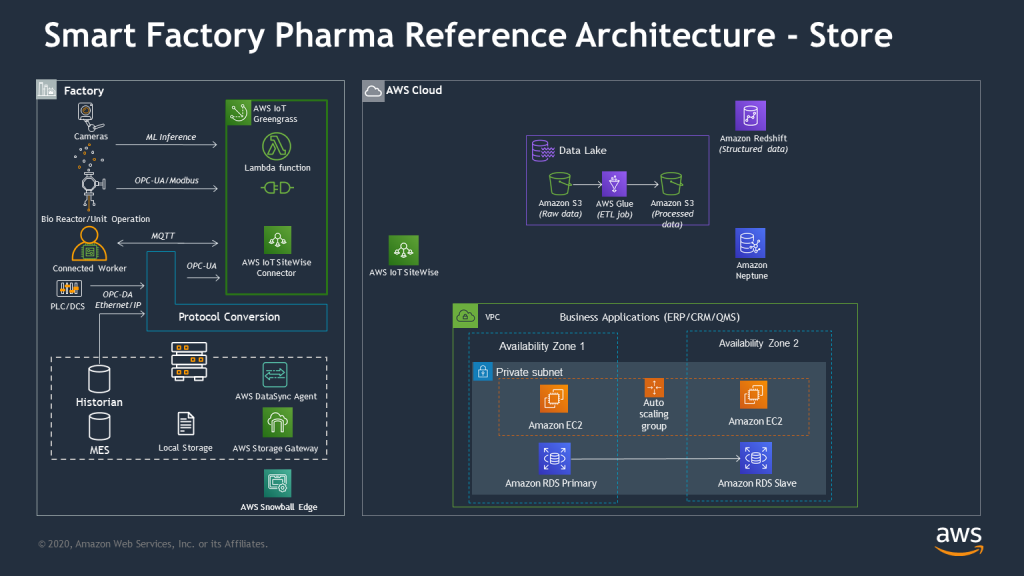
The second part of this reference architecture involves establishing cloud storage and integration.
Store: Store ensures that data silos are eliminated in an effort to drive data consolidation and access to where plant operators require visibility into plant operations. The primary technical outcome is an acute focus away from traditional on-premises infrastructure to one where hot and cold data can reside in parallel with the necessary compute resources to manage real-time shop floor operations. This pattern includes loose data coupling with business applications (ERP/CRM/QMS).
Implementing predictive analytics
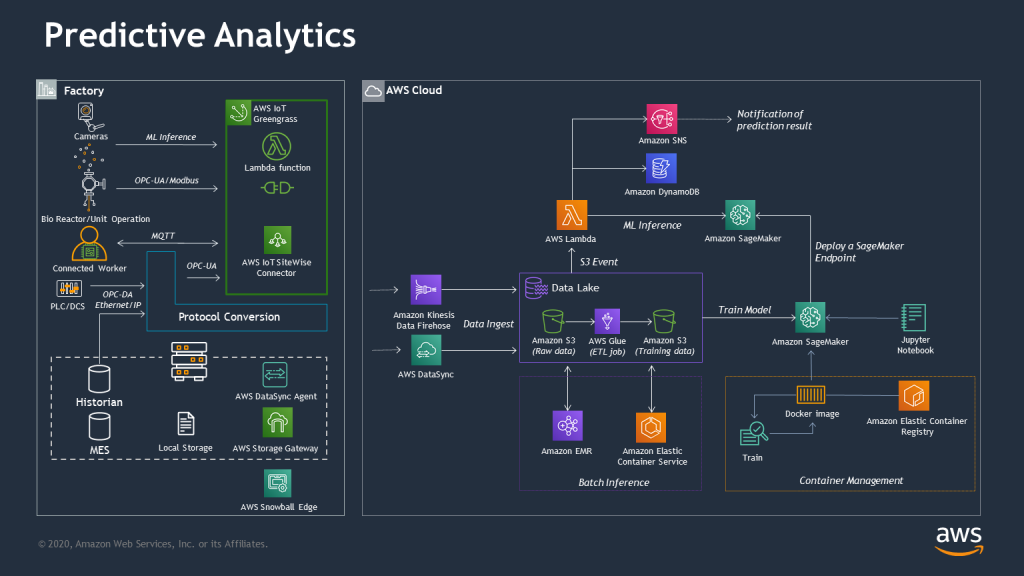
The third part of this reference architecture involves introducing new steps focusing on detection and insight generation.
Analytics: Analytics allow for rapid development and deployment of models to automate manufacturing process steps that support either real-time or retrospective workflows specific to shop floor activity. Event management from these models can be visualized through an AWS Control Tower dashboard which aggregates all shop floor events into a common multi-device supported view. The primary technical outcome to support analytics includes the use of standard, scalable services to support ETL, query, and model development. Models are then deployed either at the edge to support stream-based process steps or in the cloud to handle latency-tolerant process manufacturing state changes such as alerts, alarms, or notification events.
Alerts and real-time monitoring
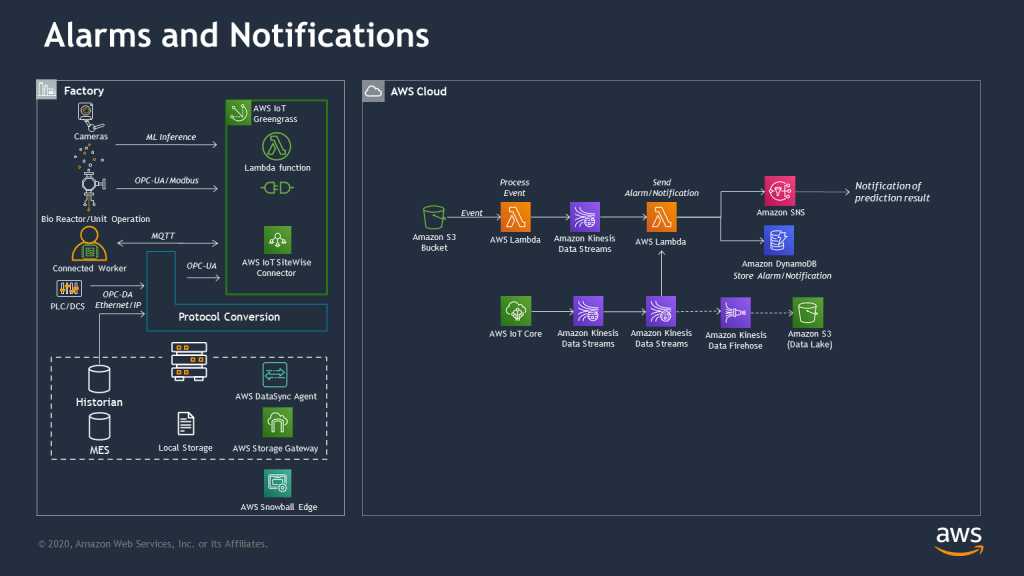
The last reference architecture phase leverages the capabilities detailed above and introduces new steps focused on proactive alerting and real-time monitoring, one of the most common use cases in manufacturing environments.
Alerts and notification: Proactively monitor dashboards and operational processes through data-driven alerts. Real-time and batch predictions generate alerts and notifications that can be pushed to customers or stakeholders or stored for further tracking and trending.
Real-time monitoring of manufacturing operations provides customers the necessary and timely information to measure and analyze events and drivers that contribute to added cost, additional waste, and reduced quality. Through real-time monitoring solutions, customers can monitor and predict negative impacts to key metrics such as throughput time, inventory levels and cost, and regulatory and quality risks.
Getting started with AWS
AWS customers such as Novo Nordisk and Merck are already innovating to create the predictive manufacturing plant. Some examples:
- Novo Nordisk uses machine learning to predict process deviations, energy usage, and equipment failures and has been able to reduce energy consumption by 20%, eliminate false rejects, and improve OEE by 1-2%.
- Merck created a data lake and uses analytics to evaluate 5.5 million batch-to-batch comparisons (15B calculations) to discover parameters driving yield improvement for vaccine production.
The bar for manufacturing innovation is quickly rising. In coming years, these approaches will transition from being innovative to being the requisite baseline of expected capabilities.
It can be challenging for life sciences customers to understand where to start, which is why AWS offers a workshop designed around manufacturing needs.
AWS conducts an objective assessment of site and network capabilities to become data-driven across several critical aspects: people, platform, operations, governance, security, and business outcomes tied to the defined business strategy.
This assessment enables customers to understand the current maturity level, agree on a target-state maturity level, and define a pragmatic roadmap to launch a set of initiatives across business and technology domains.
Another common early customer challenge is understanding how to use AWS services in a GxP environment. For more information on this topic, read the Approving AWS services for GxP Workloads post for approaches and best practices used by other life sciences manufacturers.
Reach out to your account executive to understand how you can get started with AWS to initiate or accelerate your manufacturing digital transformation.