AWS Partner Network (APN) Blog
How Implementing a Real World Evidence Platform on AWS Drives Real World Business Value
Guest post by Scot Johnson, a Solution Architect for ConvergeHEALTH by Deloitte, part of Deloitte Consulting LLP’s Innovation group (DCI).
In light of new laws such as the 21st Century Cures Act and evolving scientific insights, life sciences companies are being pressed to demonstrate clinical value to payers and health authorities. As a result, life sciences companies are shifting the way they develop and bring their pharmaceutical and medical products to market through the application of Real World Evidence (RWE). In order to discover, optimize, and demonstrate the value of RWE, life sciences companies are embracing new strategies, deeper partnerships, and innovative technology solutions. Industry-wide shifts, such as the move from volume-based to value-based payment models and more personalized healthcare, have helped fuel interest in RWE to demonstrate the value of drug and device innovations.
In this blog post, I will discuss the business drivers behind the rising importance of RWE to life sciences companies for research and product development, and how Deloitte’s ConvergeHEALTH Evidence Lifecycle Management Platform on the AWS Cloud enables RWE use cases to drive real world business value.
Delivering Value to Pharmas through RWE
The biopharmaceutical landscape has transformed due to significant advancements in science, increases in the amounts and types of data, shifts in market economics, legislation, and reimbursements. The rise of data volumes and disparate data sources, including health records, lab results, sensors, images, genomics, and claims data, have resulted in a shift from traditional research and development approaches to new collaborative models that integrate non-traditional partners across geographically dispersed resources and participants. A growing number of life sciences organizations are accommodating these disruptions with scalable on-demand storage and compute capabilities necessary to accelerate the shift toward data-driven insights and end-to-end evidence management.
Figure 1: Business value increases as RWE is leveraged across research, clinical development, and commercialization business functions
Deloitte’s ConvergeHEALTH Evidence Lifecycle Management Platform
In response to the growing demand for data-driven insights and evidence lifecycle management (ELM), Deloitte developed the ConvergeHEALTH Evidence Lifecycle Management Platform on the AWS Cloud. The ConvergeHEALTH ELM Platform leverages an integrated AWS Cloud environment preconfigured with the relevant data and tools. The ConvergeHEALTH ELM Platform consists of three main configurable layers, designed to help our clients in their efforts to quickly realize the promise of RWE and big data analytics: data layer, analysis layer, and knowledge layer. The platform’s flexible, modular design and open architectures empower domain experts and facilitate business function integration into existing and emerging plug-in analytic services from vendors and open source communities.
High-Level Architecture
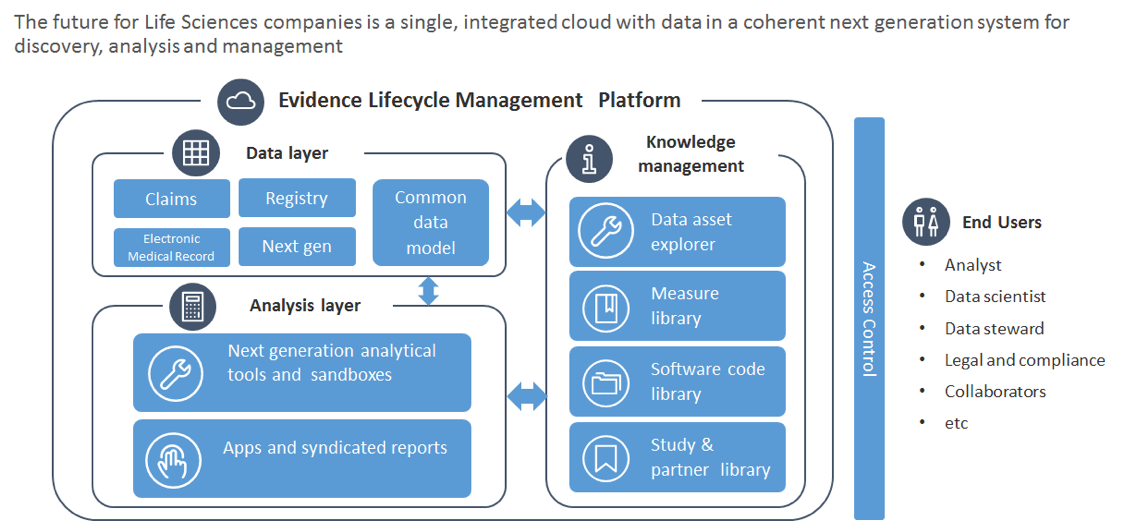
Figure 2: Reference architecture for the ConvergeHEALTH ELM Platform
Data layer – Facilitates the organization, governance, and usability of disparate datasets in support of the life science organization’s mission.
Analysis layer – Houses the analytic tools and processes for data exploration and analysis.
Knowledge management layer – Defines and powers RWE governance policies, tools, roles, and processes across the platform.
Platform Products and Accelerators
The ConvergeHEALTH ELM Platform includes several components to give enterprises visibility into information that exists within the organization and enable stakeholders to collaborate across their enterprise.
Cohort Insight – Cohort selection service (available as an API) with a web UI that allows cohort querying against datasets stored in the AWS Cloud. Allows users to iteratively apply inclusion/exclusion cohort selection criteria and facilitate interactive cohort exploration.
Cohort Integrator – Cohort synchronization service (available as an API) that synchronizes cohorts across multiple datasets, accelerating data exploration and reducing time to insights.
Data Asset Explorer & Characterization Service – A microservices API that collects domain-specific dataset profiling results for data search, discovery, and profiling.
Research Trust for Big Data – A standards-based data linkage and semantic governance model and repository built on the big data technologies Hadoop, Hive, Impala, and Spark. Research Trust facilitates the organization, linkage, semantic standardization, and exploration of data across multiple datasets.
ConvergeHEALTH Evidence Lifecycle Management Platform Implementation on AWS
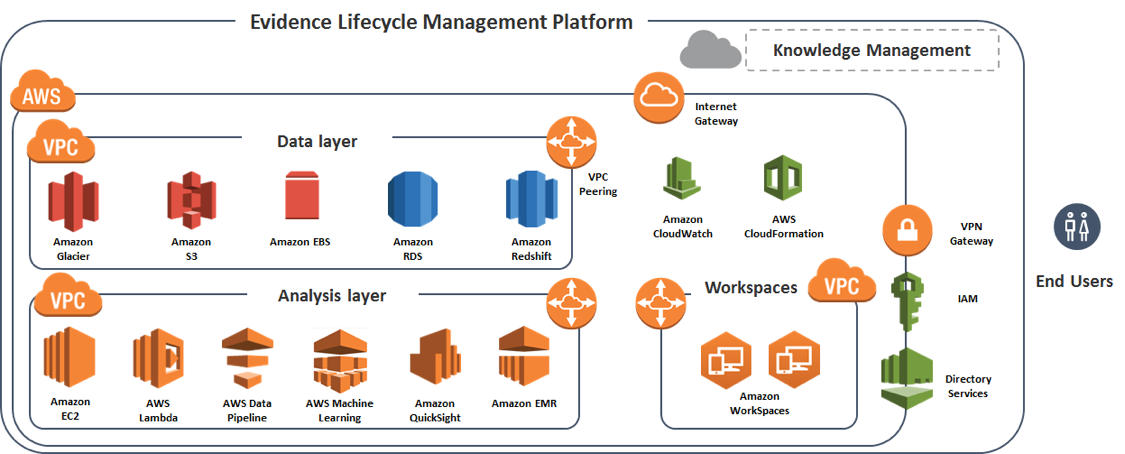
Figure 3: High-level AWS reference architecture for the ConvergeHEALTH ELM Platform
The following section highlights some key concepts of the ConvergeHealth ELM Platform and its implementation on AWS.
Security Elements
Protecting and managing patient health information (e.g., data encryption, authorized access, transporting of patient data across networks and borders) is managed by the ConvergeHEALTH ELM Platform through AWS services.
HIPAA Considerations
AWS aligns its HIPAA risk management program with FedRAMP and NIST 800-53, which are higher security standards that map to the HIPAA Security Rule. AWS provides a standard Business Associate agreement (called the “Business Associate Addendum,” or BAA) for customers deploying on the platform. Customers can use any AWS service in an account designated as a HIPAA Account under the BAA, but they must only process, store, and transmit protected health information (PHI) using certain HIPAA Eligible Services defined in the BAA.
Encryption
Consistent with the AWS BAA, PHI on the ConvergeHEALTH ELM Platform is encrypted both in-transit and at-rest. Data at-rest on Amazon Elastic Block Store (Amazon EBS) is encrypted using AWS Key Management Service (AWS KMS), and data in-transit is encrypted using [256-bit SSL].
Identity and Access Management
The AWS environment for the ConvergeHEALTH ELM Platform relies on AWS Identity and Access Management (IAM) to authorize, authenticate, and enforce user policies. IAM policies and roles allow resource access to be fine-tuned for the myriad roles within the enterprise.
Configuration Management
Virtual Private Clouds
The ConvergeHEALTH ELM Platform leverages Amazon Virtual Private Cloud (Amazon VPC), which allows customers to launch their AWS resources into a virtual network that closely resembles an on-premises environment combined with the benefits of using the scalable AWS infrastructure.
Automated Deployment
Using AWS CloudFormation templates, we’ve pre-defined and automated the deployment of the Cohort Insight application (see Figure 4). The CloudFormation template for Cohort Insight includes launch configurations and Auto Scaling groups of EC2 instances coupled with AWS Elastic Load Balancing. The template provides rapid, repeatable, and reliable on-demand deployment of Cohort Insight to handle unpredictable analytic workloads to reduce query response times and shorten time to insight.
Figure 4: AWS CloudFormation template for Cohort Insight
High Availability
AWS services have been designed to natively leverage multiple Availability Zones to build highly available, fault tolerant, and scalable solutions. For customers who desire highly available RWE applications for improved uptime necessary to meet internal stakeholders’ SLAs, the ConvergeHEALTH architecture can be configured with either a single AWS Region composed of multiple Availability Zones, or across multiple AWS Regions with multiple Availability Zones. Each Availability Zone consists of one or more discrete data centers with redundant power, networking, and connectivity.
The ConvergeHEALTH ELM Platform also uses native service features for high-availability deployment such as the Amazon Relational Database Service (Amazon RDS) and its out-of-the-box high availability and replication features.
Example Big Pharma Deployment
Vision
A Big Pharma organization was seeking opportunities to shift to value-based, personalized health care to help patients live longer, healthier lives.
Cloud Strategy
Their cloud strategy provides end-to-end visibility across the information value chain with connected processes and platforms across all functions to improve R&D productivity, product launch effectiveness, and overall operational excellence.
Solution
They chose the ConvergeHEALTH ELM Platform on AWS to serve as the global foundation for facilitating information discovery, access, analysis, governance, collaborations, and partnerships, both internally and externally.
Impact
The ConvergeHEALTH ELM Platform provides transparency into available data across the enterprise, facilitating the sharing of insights and enabling researchers to collaborate. Multiple disparate datasets are connected to gain new insights. And by developing new coordinated partner strategies, they are able to build networks of strategic partners with advanced and integrated expertise.
Realizing the Business Benefits from RWE and Next Steps
Managing the shift from volume-based to value-based payment models and the move to personalized health care will require more agile real-world evidence capabilities along with new strategies, partnerships, and technologies. Pharmas that manage the transformative shift to operationally utilize RWE will be positioned to realize the potential and business value of RWE. Deloitte focuses on helping companies achieve these results/objectives through its relationship with AWS and the blending of our skills and capabilities to make our client’s transformative shift to RWE a manageable journey.
To learn more about the ConvergeHEALTH Evidence Lifecycle Management Platform, see the ConvergeHEALTH website.
About Deloitte
Deloitte professionals guide traditional health care and life sciences companies and new market entrants in navigating the complexities of the US and global health care system. As market, political, and legislative changes alter the industry, we help our clients develop innovative and practical solutions.
As used in this document, “Deloitte” means Deloitte Consulting LLP, a subsidiary of Deloitte LLP. Please see www.deloitte.com/us/ about for a detailed description of our legal structure. Certain services may not be available to attest clients under the rules and regulations of public accounting.
The content and opinions in this blog are those of the third party author and AWS is not responsible for the content or accuracy of this post.

