AWS for Industries
Finding enduring solutions to the evolving COVID-19 crisis
Blog guest authored by Bo Barnhart, Vice President of Translational Research at AbCellera
In February 2020, AbCellera mobilized its pandemic response platform to find treatments for COVID-19. Supported by Amazon Web Services (AWS), AbCellera discovered two antibody treatments, bamlanivimab and bebtelovimab, for COVID-19 in a year, breaking records to keep pace with an evolving pandemic.
At the beginning of the pandemic, vaccines and drugs designed to target SARS-CoV-2 were extremely limited. Millions worldwide were left vulnerable to the new viral threat and had to rely on drugs that managed symptoms rather than stopping the virus from infecting cells. Antibodies can be powerful tools to treat and prevent the spread of infectious diseases. These Y-shaped proteins form a central part of the immune systems’ infection-fighting arsenal and can be developed into treatments faster than small molecule drugs such as antivirals.
Digging deeper into the immune database
Finding the antibodies that most effectively block a virus can be challenging because humans have billions of antibodies circulating in their immune systems. Each antibody-producing immune cell has to be captured and analyzed individually, creating massive, multidimensional data that are too complex for traditional spreadsheets to handle. Viruses also change over time, so technology for finding antibodies needs to be powerful, fast, and flexible to keep pace with an evolving pandemic.
Conventional workflows for finding antibodies aren’t built for speed. They can only analyze a small fraction of the immune system at a time. In addition, fragmented approaches result in data loss at each step, limiting the chances of finding the most promising antibodies to develop into drugs.
AbCellera’s solution to this problem is an integrated technology stack that searches, decodes, and analyzes natural immune systems to find antibodies that can be developed into treatments. AbCellera’s integrated data infrastructure, coupled with AWS’ on-demand scalability, allowed AbCellera’s technology stack to process the massive amounts of data generated during its pandemic response.
These capabilities enabled AbCellera to move fast and discover bamlanivimab – the first COVID-19 antibody treatment to be authorized by the U.S. FDA. As the pandemic evolved, and new variants emerged, AbCellera used its platform again to identify a second antibody, bebtelovimab, that is broadly effective against all known variants of concern including the Omicron BA.2 subvariant.
“Biology is a data science,” says Carl Hansen, Ph.D, CEO of AbCellera. “We need technology firms that are investing in solutions to make the process faster and to sharpen the ax of drug discovery.”
Breaking records to develop a COVID-19 treatment
AbCellera has been developing a platform to respond rapidly to viral outbreaks since 2018 as part of the Defense Advanced Research Projects Agency (DARPA) Pandemic Prevention Platform (P3) program. Within a week of receiving a sample from one of the first patients in North America to recover from COVID-19, AbCellera screened over five million antibody-producing cells, and found around 500 unique antibodies, the largest panel of human antibodies against SARS-CoV-2 to be reported at the time.
“We were able to substantially expand our on-premise analytical capacity by using AWS, using 10 times the amount of computing power with very little effort,” says Rodrigo Goya, Ph.D, Bioinformatics Lead at AbCellera. “It was very useful to have all the data in Amazon S3 buckets and be able to access it from base to nodes without having to deal with file systems and all the related infrastructure.”
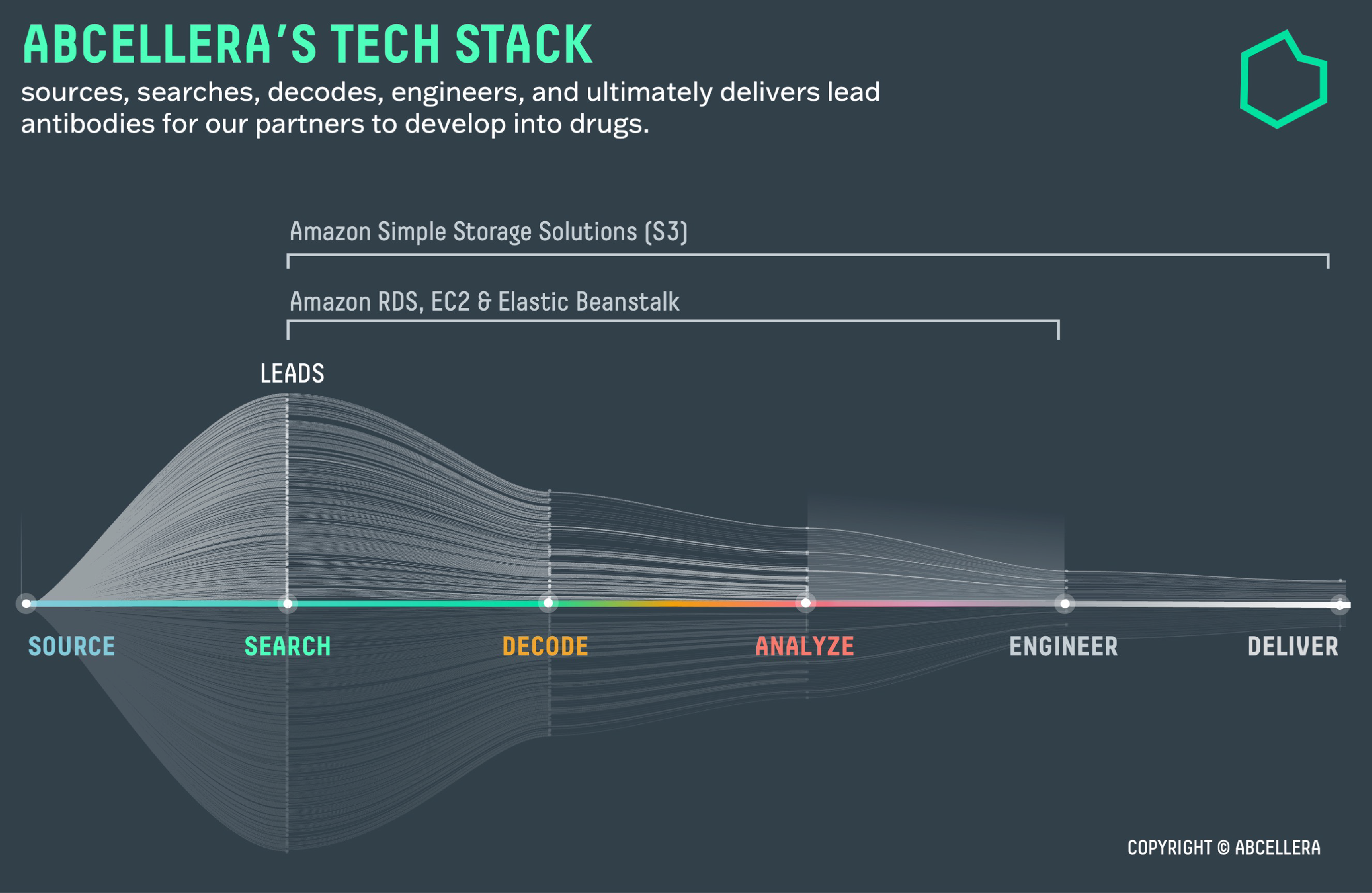
To select lead candidates, AbCellera assessed each of these 500 antibodies across 500 different parameters. This process generated more than 250,000 data points. To store and organize its data, AbCellera used Amazon Simple Storage Service (Amazon S3) and Amazon Relational Database Service (Amazon RDS) (see Figure 1) because it offers industry-leading scalability, data availability, security, and performance.
Using Celium, AbCellera’s proprietary computational engine for antibody discovery, teams visualized and analyzed this large dataset. Working cooperatively to filter this dataset on Celium, which leverages Amazon EC2 instances, they identified 24 lead candidate antibodies in 23 days.
“During the COVID-19 pandemic where every hour mattered, access to AWS was so important,” says Maia Smith, Head of UI/UX Design. “With AWS, multiple scientists can access Celium simultaneously, collaborating to determine which antibodies should move into the next stages of clinical development.”
This campaign led to the discovery of bamlanivimab, which entered human testing 90 days after AbCellera launched its COVID-19 response. Since then, bamlanivimab has helped more than one million patients, keeping over 100,000 people out of the hospital and saving over 40,000 lives.
Figure 1 – AbCellera’s Tech Stack
Taking aim at moving targets
As the pandemic persisted, a new challenge arose. Dangerous viral variants that spread more easily and evaded available COVID-19 treatments quickly gained dominance, once again leaving millions at risk. To keep pace with this viral evolution, AbCellera had to dive deeper into the human immune response in search of ultra-rare, broadly-effective antibodies that could protect against SARS-CoV-2 variants. AbCellera turned to Amazon EC2 and AWS Elastic Beanstalk (Elastic Beanstalk) to accommodate its increased demand for multi-user data access, storage, and analytical bandwidth.
“We can focus on the science of finding antibodies rather than having to devote resources into scaling our computing infrastructure,” says Jordan Kowalski, Associate Director of Data Software at AbCellera.
From this effort AbCellera identified bebtelovimab, an antibody that disarms all known SARS-CoV-2 variants of concern (including the Omicron variant and subvariants) by binding to a rarely-altered part of the virus. Bebtelovimab is at least 50 times stronger than other antibodies that are authorized or in clinical development against the Omicron variant, and it is the only antibody that retains its strength against the quickly-spreading BA.2 subvariant. Bebtelovimab was given emergency use authorization by the U.S. FDA in February 2022 and is currently being used to treat patients across the U.S.
Figure 2 – AbCellera’s co-developed two antibody treatments authorized by the U.S. FDA to treat COVID-19
Staying one step ahead
Outside of its pandemic response, AbCellera is working on more than 150 drug programs that span oncology, pain, neurodegeneration, infectious disease, autoimmune disease, allergic inflammation, ophthalmology, women’s health, and cardiovascular disease.
AbCellera continues to invest heavily into its technology stack, and plans to integrate additional AWS services into its growing pipeline. Hansen says, “We believe that if we can put the best science and the best innovators in touch with the best technologies, we’ll get drugs to patients faster, and everyone wins.”
 Bo is Vice President of Translational Research at AbCellera where he works with partners to evaluate, optimize, and nominate developable lead antibody candidates generated by AbCellera. Bo has over 12 years of antibody drug development experience and has progressed more than ten therapeutics through early clinical development, with several entering first-in-human testing. He completed his PhD in Immunology at the University of Chicago and his BA in Biology and English Literature at Rutgers University, and a post doc from University of Penn. Outside of work, he spends as much time as possible with his family, gets outside for hikes, and plays tennis.
Bo is Vice President of Translational Research at AbCellera where he works with partners to evaluate, optimize, and nominate developable lead antibody candidates generated by AbCellera. Bo has over 12 years of antibody drug development experience and has progressed more than ten therapeutics through early clinical development, with several entering first-in-human testing. He completed his PhD in Immunology at the University of Chicago and his BA in Biology and English Literature at Rutgers University, and a post doc from University of Penn. Outside of work, he spends as much time as possible with his family, gets outside for hikes, and plays tennis.