AWS Startups Blog
Changing the Landscape of Molecular Testing
Guest post by Paul Flook, PhD, CIO and VP, Software Engineering, ChromaCode
This past year has demonstrated, now more than ever, the critical need to be able to develop and deploy rapid molecular testing at scale. The ability to do this has emerged as a major differentiator for ChromaCode, a startup diagnostics company based in Carlsbad, California. We focus on developing Real-Time Polymerase Chain Reaction (RT-PCR) tests that can detect multiple genes or pathogens in biological samples.
Before the onset of COVID-19, our focus had been on tackling other important challenges such as tick-borne pathogens and drug resistant bacterial genes using our proprietary High-Definition PCR (HDPCR™️) platform1. Our unique advanced multiplex technology is well-suited for addressing the ongoing research needs due to rise of tick-borne diseases or the 2.8 million cases of antibiotic-resistant infections in the US per year. But testing for these targets has taken a back seat as most nations have been consumed with COVID-19 testing.
In the spring of 2020, just as the scale of the pandemic was becoming clear, ChromaCode worked with a lab at University of California San Diego to prototype a COVID-19 molecular test2. As this progressed and we saw the problems faced by labs ramping up their development efforts, we realized there was a significant opportunity for us to help with COVID-19 testing. A critical enabler in the resulting effort was the design of our software, ChromaCode Cloud™, which made it possible to quickly deliver the data interpretation and reporting components of the test. A working assay was ready within about a week from the start of the project, and we are proud to have since shipped more than four million COVID-19 tests in less than eight months, despite the numerous roadblocks that inherently plague diagnostic testing.
Rapid roll out of new molecular tests is a key challenge
Labs don’t have the time to validate, install, and maintain new software and hardware, and most organizations strive to keep their investment in these to a minimum. Together these can represent significant barriers to adoption of new diagnostics in labs seeking to offer the best tests at the lowest cost.
In addition to the technology itself, there tend to be various challenges in delivering a solution that can be easily integrated into a lab’s infrastructure. There are compliance and data privacy issues with diagnostic products. Maintaining compatibility with critical operating system updates may require updates that customers need to install and validate themselves. But research labs cannot spend all their time certifying software updates. The list goes on.
A unique cloud-based solution for medical devices
ChromaCode’s HDPCR™️ platform changes the conversation around molecular testing. The technology supports a simple workflow that enables users to upload standard PCR instrument output files to our software platform using a web application. Data is automatically analyzed and results reported to the end user. Providing a 4x expansion of the analytical capacity of most PCR instruments, labs can leverage their existing PCR instruments, maintain their existing protocols, and can scale at a higher rate.
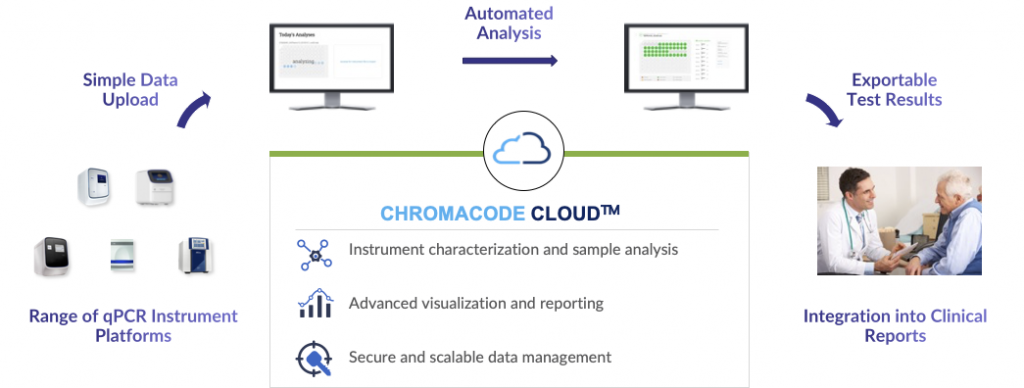
Our technology uses digital signal processing, enhanced PCR reagents, and cloud-based analytics. It’s this unique combination that enables us to significantly increase test capabilities while decreasing costs, factors that contributed to being named as a Main Track Winning Team in the XPRIZE Rapid COVID Testing competition. Over 100 entrants participated in this competition which was designed to recognize the best low cost, high throughput COVID tests. Our success emphasized how ChromaCode’s patented technology is changing the PCR landscape, and how ChromaCode Cloud enables increased scale.
ChromaCode Cloud is comprised of a web application, analysis tools, and multiple platform services running in a Kubernetes cluster and using AWS foundational products such as Amazon EC2, Amazon RDS, and Amazon S3 to deliver an end-to-end analysis solution. Our use of AWS from the start avoided the need for us to build and operate our own core infrastructure and allowed us to rapidly scale as the COVID testing demand exploded. We have also built a separate informatics platform that enables us to analyze aggregate de-identified analysis and operations data to gain insights into the performance of our tests.
Because of this, with nominal cost deployment, we can better support our customers with the cloud, which leverages the agility, security, breadth and depth of AWS services to enable real-time access to the data required to support a wide range of accurate and affordable molecular diagnostics, including COVID-19 testing.
The high availability of the system means we can deploy new maintenance and security updates immediately without disruption to customers; there’s no need to stop testing or revalidate security updates, for example. With effectively unlimited storage capacity, we can free customers from concerns about data archiving. In terms of compliance and security standards, AWS has industry-leading capabilities, superior to anything a typical customer would implement themselves.
By leveraging a cloud-based software solution, not only are we able to provide an improved experience, but labs are able to easily adopt and rapidly scale. And, we now have an incredibly rich data set. We are currently analyzing aggregated de-identified data that allows us to look at the performance of our assays, gain better insights into our inventory, and even offers us the potential to look at epidemiological trends.
Expanding our testing capabilities to address new challenges
The ongoing challenges around COVID-19 mean that an important next step for ChromaCode is an expansion of our testing capabilities to support new demands on labs at the frontline. A primary objective has been to develop a new test that can identify additional respiratory viruses such as the seasonal flu and Respiratory Syncytial Virus (RSV), whose symptoms may resemble COVID-19. We are currently planning the extensive clinical trials necessary for this new test to be cleared by the FDA as a full In Vitro Diagnostic test.3
We are also developing research use only tests to identify new COVD-19 variants3. The emergence of new strains that have different etiologies and transmissibility pose significant risks to controlling the pandemic. Genomic sequencing has been used effectively to characterize new strains, but it is impractical to apply this at population scale. Instead, rapid development of PCR-based screening assays and tests for variants may be critical to understand large scale epidemiological changes.
Not only is ChromaCode Cloud™ imperative to support these tests, but our ability to aggregate de-identified test data in our informatics platform also means that we are able to undertake large scale monitoring of different variants that can provide unique insights.
If you wish to learn more about ChromaCode, our products or our software you can visit chromacode.com, or request a copy of our white paper from info@chromacode.com.
1 For Research Use Only. Not for use in diagnostic procedures.
2 For Emergency Use Authorization Only. For in vitro diagnostic use. For prescription use only.
3 The content provided herin may relate to products or workflows that are still in development, have not been officially released or fully validated and is subject to change without further notice.