AWS for Industries
Transforming site monitoring in clinical trials
Clinical trial monitoring is an integral part of running a successful and compliant clinical trial. Currently the site monitoring process is manual, costly, and subject to data quality issues. This post discusses how various aspects of site monitoring can be automated to optimize cost and increase quality. About 25% to 30% of the total cost of a clinical trial goes into monitoring. More importantly, effective monitoring enables better clinical outcomes. The COVID-19 pandemic has increased the urgency to move toward a centralized remote monitoring option. This will accelerate the speed of the clinical trial process while improving efficiency and reducing costs.
Monitoring ensures the following:
- Human rights of patients involved in the clinical trials are protected by making sure that trial protocols are adequately followed and data relating to various aspects of the trial like adverse events and dosage information is collected.
- Data collected and submitted by the clinical trial site is accurate.
- The site complies with all Food and Drug Administration (FDA) regulations relating to good clinical practice (GCP) and clinical trials.
To ensure the human rights of the patients involved, a site monitor, the Clinical Research Associate (CRA), performs various checks that are currently done manually and on site. For example, the site monitor checks all Informed Consent Forms (ICFs) to ensure they have been duly signed by the patient as a verification that informed consent forms have been read and understood by patients. The monitor also verifies the eligibility criteria and ensures that all subjects (patients) are eligible for the trial. The monitor makes sure that all the adverse events are being reported by the site by verifying the source systems and the reports.
Accuracy and completeness of data in a clinical trial is of paramount importance not just from a safety and efficacy end point perspective but from a regulatory and compliance standpoint. Site monitors play a key role in this. The monitors must verify the accuracy and completeness of the case report forms with respect to the source documents and any cross references among documents. They must also verify the dose and therapy modifications done for specific patients are accurately documented.
From a regulatory perspective, monitors are responsible for ensuring that sponsors have provided investigative brochure to each site. They have to verify the qualifications of the investigators. Monitors also confirm if the investigator and staff are adequately informed about the trials and trained on the protocol.
Site monitors spend about 20% of their time moving between sites and another 20% verifying different documents. At the same time, they communicate with the investigator and staff to ensure the protocol is followed and any deviations are appropriately captured and reported to the sponsor for further action. Monitors should also communicate any deviations from protocol, standard operating procedures (SOPs), GCP, and the regulations to the investigator.
Monitors submit a written report to the sponsor after each visit. Reports cover the following:
- Summary of what was reviewed
- Any significant findings
- Actions taken
- Recommendations
AWS services can be used in a design pattern to enable the digital transformation in this space. The objective is to automate the cross verification of documents and flag exceptions for the site monitor to review, intervene upon, and resolve. This reduces errors due to manual verifications and improves the quality of the data. That, in turn, improves the safety of patients enrolled in a trial.
In capturing this data and using artificial intelligence (AI) and machine learning (ML), the solution can flag sites that are risk prone based on past deviations in clinical trial protocols. Monitors can then effectively use their time by focusing on those sites. This type of analysis provides a risk profile for the sites to be leveraged for future such trials.
Reference Architecture
The following subsections detail the specific parts of the Reference Architecture.
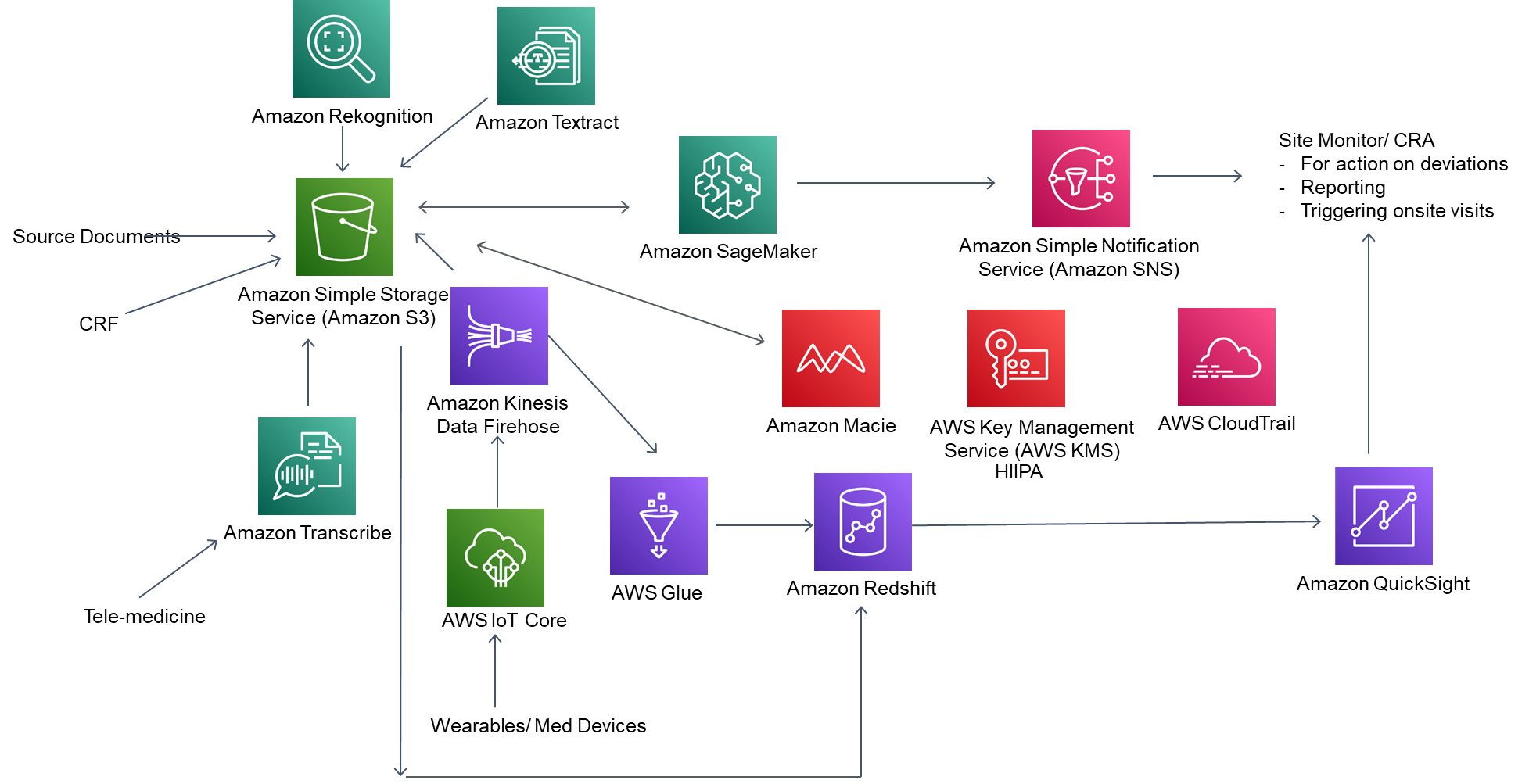
Data Collection
With Amazon Textract, you can read text from scanned handwritten documents and store it (by patient) in Amazon Simple Storage Service (S3) buckets. Optionally, you can leverage Amazon Elastic File System (Amazon EFS) for multiple patient files from multiple locations. For informed consent, you can leverage Amazon Rekognition to verify signatures with a valid identity. You can store electronic health record (EHR) data in S3 buckets. You can use AWS IoT Core for data collection from wearables through Amazon Kinesis firehose into Amazon S3.
Data Transcription
For doctor–patient conversations, you can leverage Amazon Transcribe Medical to convert speech to text and store the data in Amazon S3.
Confirmations
The monitor must confirm whether the information brochure has been provided to the investigator’s office. This can be done by centrally storing the brochure, emailing a link, and getting read confirmations from people involved in trails at the investigator’s office. Any deviations can be reported and followed up.
Similarly, the monitor must confirm if the staff in the investigator’s office are aware of the trials. This can be done through proctoring via video recording with an upload of identification document. Amazon Rekognition, Amazon Transcribe Medical, and Amazon SageMaker can do the verifications and log the details or send it for action to the monitor if required.
Identifying Source Data Deviations
By leveraging SageMaker, you can create learning algorithms to analyze the data, identifying deviations among source documents and between the source documents and the case report form (CRF) and sending messages to the site monitor with details for further investigation and reporting.
SageMaker can identify missing source documents and send alerts to both the investigator and the monitor. Based on patterns, the algorithm can also suggest targeted onsite monitor visits while keeping the sponsors informed of any emerging deviation patterns among various sites. This provides a thorough risk-based and high-quality approach at a reduced cost.
Data Analytics
Bringing clinical trial data together in Amazon S3 has significant advantages not just for clinical trial monitoring, but for the trial as a whole. While Amazon S3 will store the structured and semi-structured data, it can be analyzed to create structured data sets by leveraging Amazon Glue to load data into Amazon RedShift for data warehousing and querying. Amazon Quicksight can be used by sponsors and monitors for querying data.
HIPAA Compliance and Good Clinical Practice
Securing and encrypting patient data is essential as part of compliance. The architecture leverages AWS Key Management Service (AWS KMS), Amazon Macie, and AWS CloudTrail for encryption, monitoring personally identifiable information (PII) data, and having full traceability. Further, data quality, real-time reporting, and predictive analytics for risk assessment of various trial sites ensure good clinical practice (GCP).
The various clinical trial source documents and any wearables data are collected in Amazon S3. Amazon Rekognition will enable image verification (like verifying signatures). AI/ ML models built in Amazon SageMaker will help identify deviations in source documents and will flag it to the monitors via Amazon SNS. Amazon Glue can extract the data from Amazon S3 and transform the data for loading into Amazon Redshift to be ready for dashboards for various user personas. These dashboards will also provide risk profile analysis for various clinical trial sites. Amazon Macie and Amazon Cloud Trail will provide the security and compliance for this solution.
Conclusion
The current situation with COVID-19 creates an urgent need to help pharmaceutical companies and Contract Research Organizations (CROs) move towards a centralized risk-based hybrid (remote plus targeted onsite) monitoring approach. This approach uses technology to automate many of the cumbersome and manual functions performed by site monitors and help them become more effective and efficient by focusing their time on the quality of the trials. This automation also eliminates errors and improves the data quality and therefore the safety of clinical trial patients. This is a perfect opportunity to use available AWS cloud technology and enable a significant digital transformation in this critical area of clinical trials.